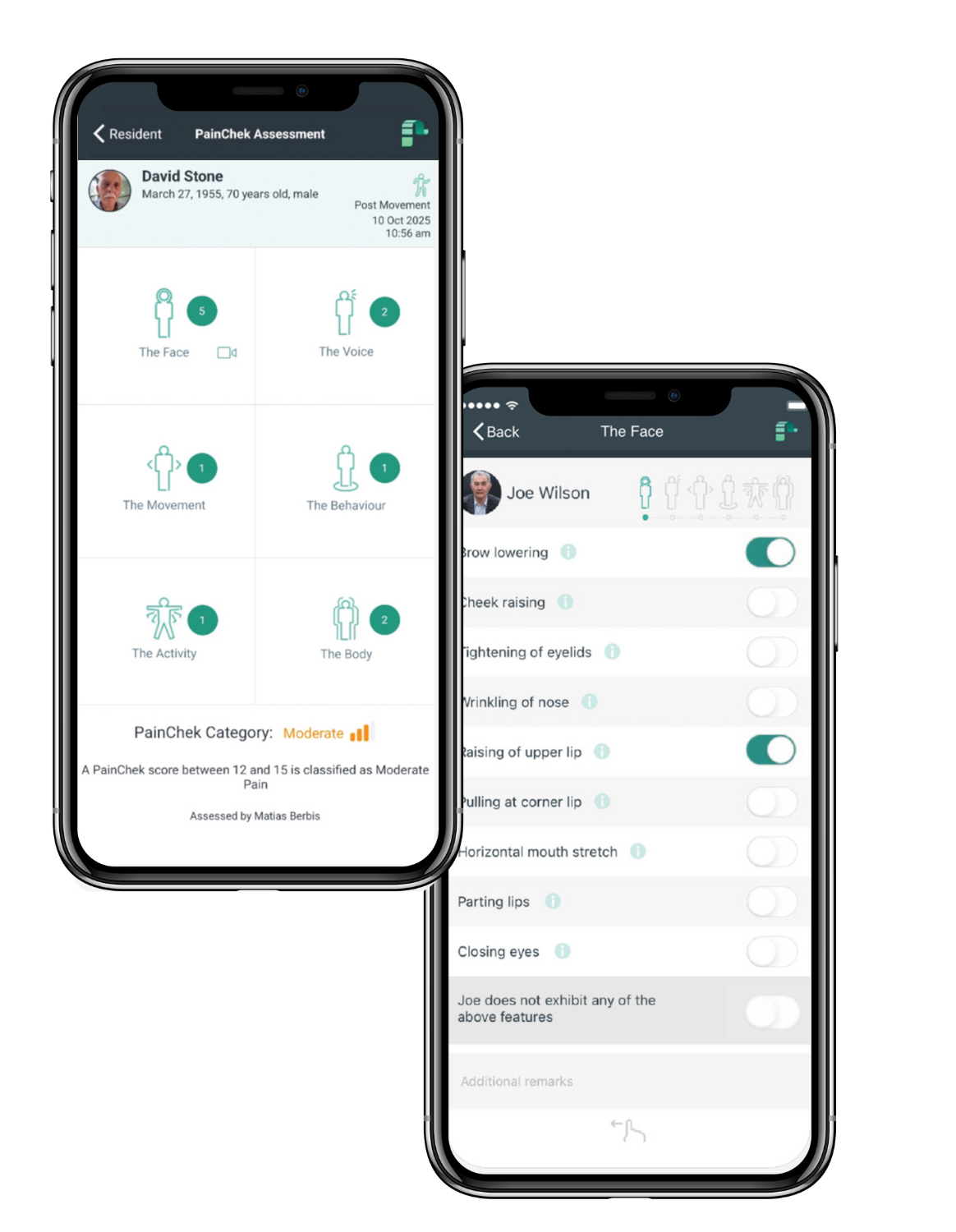
PainChek® uses evidence-based AI technology to automatically detect facial micro-expressions that indicate pain, along with digital checklists to record carer observations of other pain-related features.
1. Choose the assessment method
- Use AI-driven facial analysis for those who cannot reliably verbalize pain.
- Use the Numerical Rating Scale (NRS) for those who can
self-report. - For individuals with fluctuating communication abilities, use one or both methods as appropriate.


2. Capture observations
- Record facial expressions, movement, vocalizations, and other pain-related behaviors through the guided PainChek® framework.
- For self-report, ask the guided NRS question and document the response, along with time, date, and activity status (‘at rest’ or ‘post movement’).
3. Determine pain intensity
PainChek® analyzes the data and classifies the individual’s pain intensity as no pain, mild pain, moderate pain, or severe pain, combining AI detection and observational/self-report inputs as needed.


4. Store and track data
All results are securely stored in the PainChek® cloud portal for continuous monitoring of pain trends and treatment effectiveness.
5. Guide care decisions
Use the pain intensity classification and trends to inform personalized interventions, monitor effectiveness, and support clinical decision-making at the point of care.

AI-enabled insights for better pain management
Detailed reports from PainChek® Analytics provide actionable insights, helping healthcare professionals understand how pain is experienced and tailor a personalized approach to care.

Interested in implementing PainChek in your facility?
Seamless integration options
PainChek® connects effortlessly with your eMAR and EHR systems via open APIs. Data flows both ways: resident information updates automatically, and time-stamped pain assessments are delivered instantly.
No duplicate work, no manual entry – just accurate pain insights at the point of care.


Validated in pivotal US clinical trial
With no comparable device on the US market, PainChek® underwent the FDA’s De Novo review, requiring evidence of safety and effectiveness to establish a first-of-its-kind, new category medical device.
Results from the US Validation Study showed PainChek® demonstrated strong agreement with the internationally recognised Abbey Pain Scale, delivering accurate pain category assessments and superior test-retest reliability. It performed consistently across different resident demographics (age, gender, race, and ethnicity) and device platforms, with statistical validation further confirming its accuracy and clinical strength.
Proven global outcomes
Across multiple studies and care settings in Australia, New Zealand, the UK, and Canada, PainChek® has consistently delivered measurable clinical outcomes:
- Medication optimization: Reduced use of analgesics, antipsychotics, antidepressants, anxiolytics, and hypnotics
- Resident well-being: Improved nutrition, increased mobility, reduced dependency, and improved quality of life
- Safety and care quality: Fewer falls, reduced distress, and improved staff confidence in pain assessment
- Operational benefits: Improved care efficiency and significant cost savings



























